

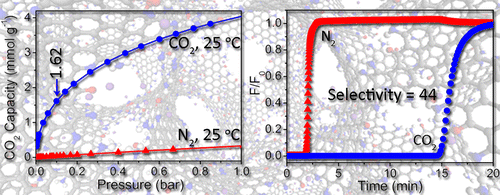
 We designed and prepared a novel microporous carbon material (KNC-A-K) for selective CO2 capture. The combination of a high N-doping concentration (>10 wt %) and extra-framework cations, which were introduced into carbonaceous sorbents for the first time, endowed KNC-A-K with exceptional CO2 adsorption capabilities, especially at low pressures. Specifically, KNC-A-K exhibited CO2 uptake of 1.62 mmol g–1 at 25 °C and 0.1 bar, far exceeding the CO2 adsorption capability of most reported carbon material to date. Single component adsorption isotherms indicated that its CO2/N2 selectivity was 48, which also significantly surpasses the selectivity of conventional carbon materials. Furthermore, breakthrough experiments were conducted to evaluate the CO2 separation capability of KNC-A-K on CO2/N2 (10:90 v/v) mixtures under kinetic flow conditions, and the obtained CO2/N2 selectivity was as high as 44, comparable to that predicted from equilibrium adsorption data. Upon facile regeneration, KNC-A-K showed constant CO2 adsorption capacity and selectivity during multiple mixed-gas separation cycles. Its outstanding low-pressure CO2 adsorption ability makes KNC-A-K a promising candidate for selective CO2 capture from flue gas. Theoretical calculations indicated that K+ ions play a key role in promoting CO2 adsorption via electrostatic interactions. In addition, we found that HCl molecules anchored in N-doped carbon have a similar promotion effect on CO2 adsorption, which contradicts the conventional wisdom that the neutralization of basic sites by acids diminishes the adsorption of acidic CO2 gas.
We designed and prepared a novel microporous carbon material (KNC-A-K) for selective CO2 capture. The combination of a high N-doping concentration (>10 wt %) and extra-framework cations, which were introduced into carbonaceous sorbents for the first time, endowed KNC-A-K with exceptional CO2 adsorption capabilities, especially at low pressures. Specifically, KNC-A-K exhibited CO2 uptake of 1.62 mmol g–1 at 25 °C and 0.1 bar, far exceeding the CO2 adsorption capability of most reported carbon material to date. Single component adsorption isotherms indicated that its CO2/N2 selectivity was 48, which also significantly surpasses the selectivity of conventional carbon materials. Furthermore, breakthrough experiments were conducted to evaluate the CO2 separation capability of KNC-A-K on CO2/N2 (10:90 v/v) mixtures under kinetic flow conditions, and the obtained CO2/N2 selectivity was as high as 44, comparable to that predicted from equilibrium adsorption data. Upon facile regeneration, KNC-A-K showed constant CO2 adsorption capacity and selectivity during multiple mixed-gas separation cycles. Its outstanding low-pressure CO2 adsorption ability makes KNC-A-K a promising candidate for selective CO2 capture from flue gas. Theoretical calculations indicated that K+ ions play a key role in promoting CO2 adsorption via electrostatic interactions. In addition, we found that HCl molecules anchored in N-doped carbon have a similar promotion effect on CO2 adsorption, which contradicts the conventional wisdom that the neutralization of basic sites by acids diminishes the adsorption of acidic CO2 gas.